Enthalpy of neutralization of the reaction between CH3COOH(aq) and NaOH(aq) is - 13.2 kcal/eq and that of the reaction between H2SO4(aq) and KOH(aq) is - 13.7 kcal/eq . The enthalpy of dissociation
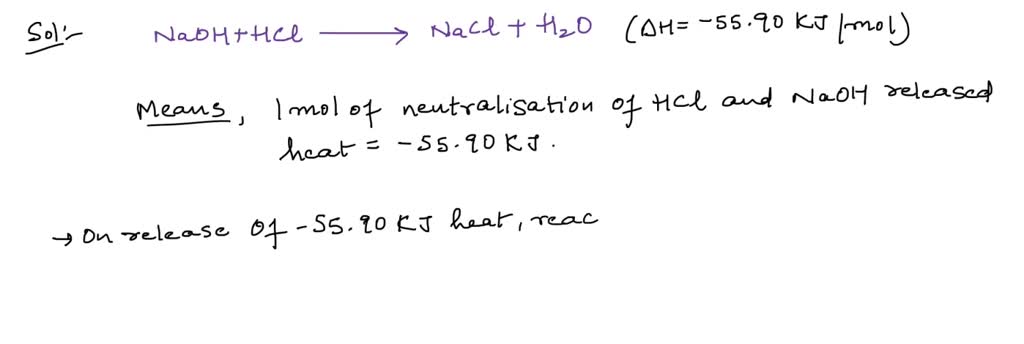
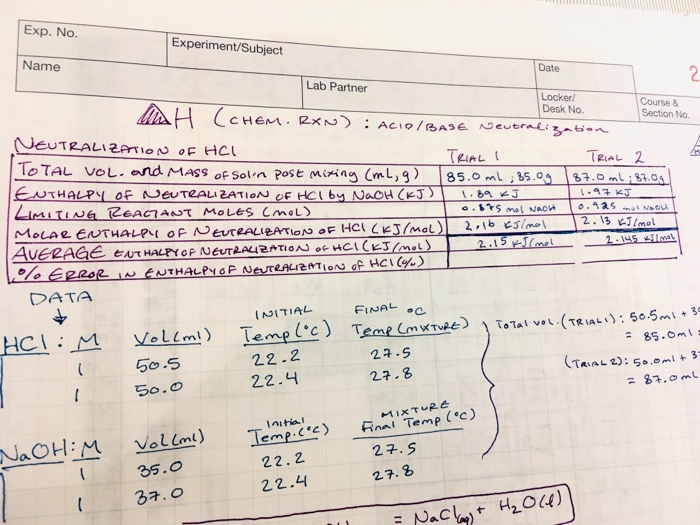
SOLVED: In calculating the enthalpy of neutralization between HCl and NaOH. If the concentration of HCL was actually 5% higher than that of what was used in the original calculation what would

If enthalpy of neutralization of HCl by NaOH is - 57kJmol^-1 and with NH4OH is - 50kJmol^-1 , then calculate the enthalpy of ionisation of NH4OH(aq) .

The Enthalpy of Neutralization of Phosphoric Acid Worksheet - Make sure to include the relevant - Studocu