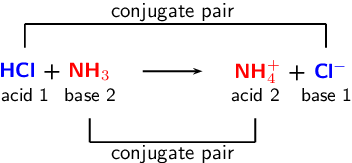
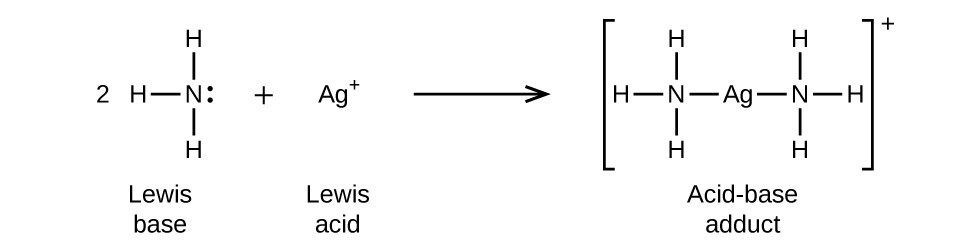
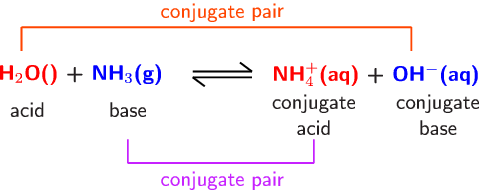
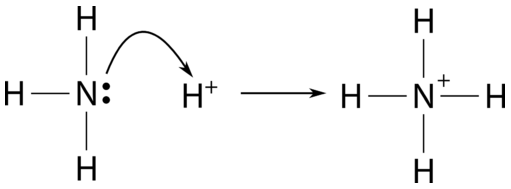
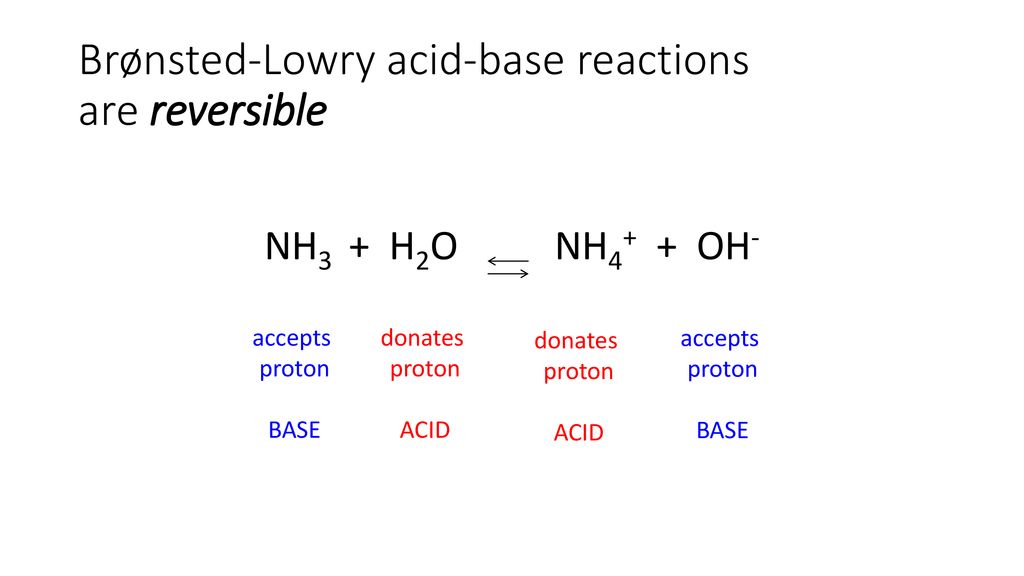
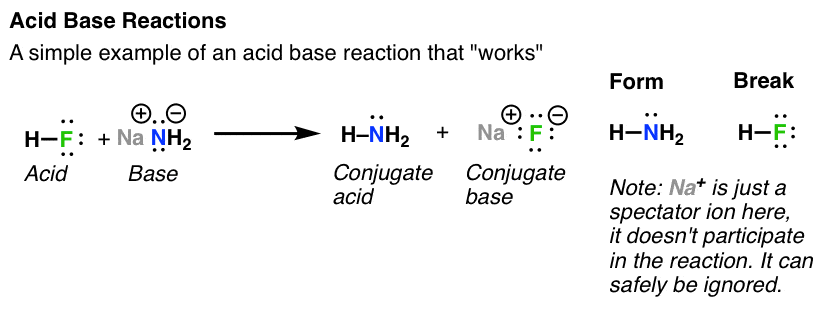
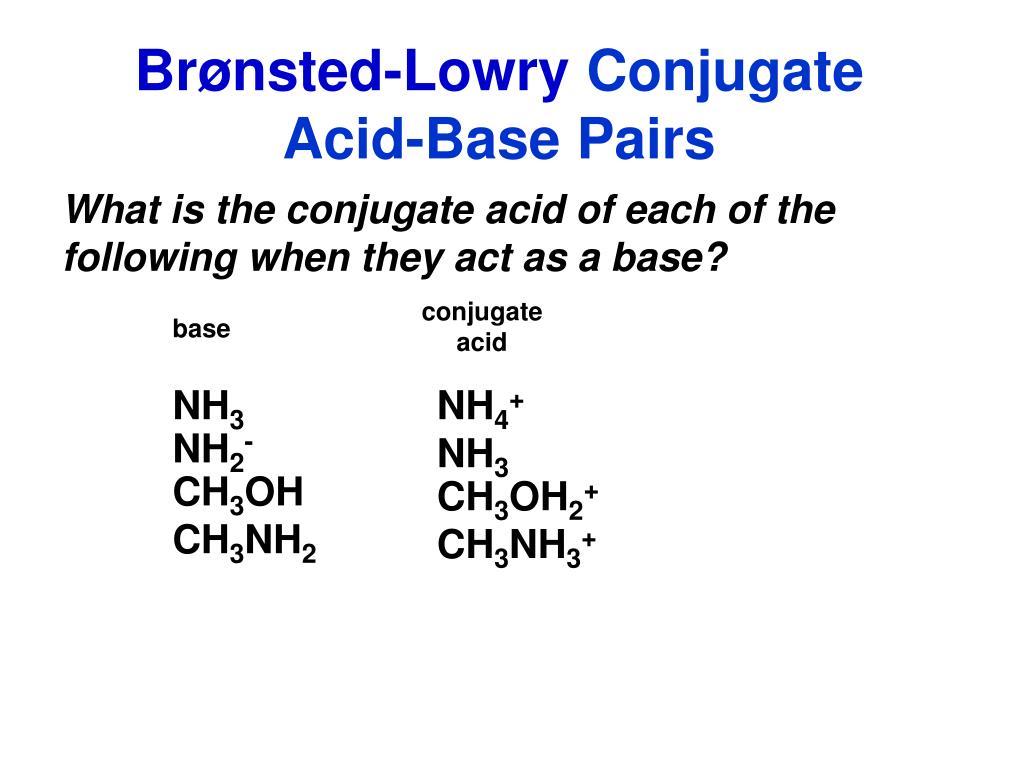
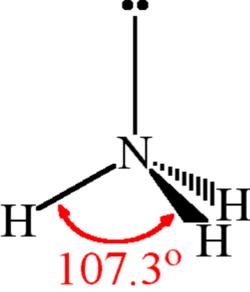
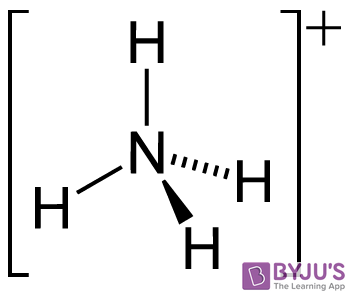
SOLVED: Identify the conjugate acid and base pairs in the following acid base reaction: HCO3 NH3 CO3-2 NH4t Acidlconjugate base: HCO3 CO3-2 Baselconjugate acid NH3 NH4 B Acidlconjugate base: CO3-2 / HCO3"

Identify the conjugate acid-base pairs in this equilibrium. NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com