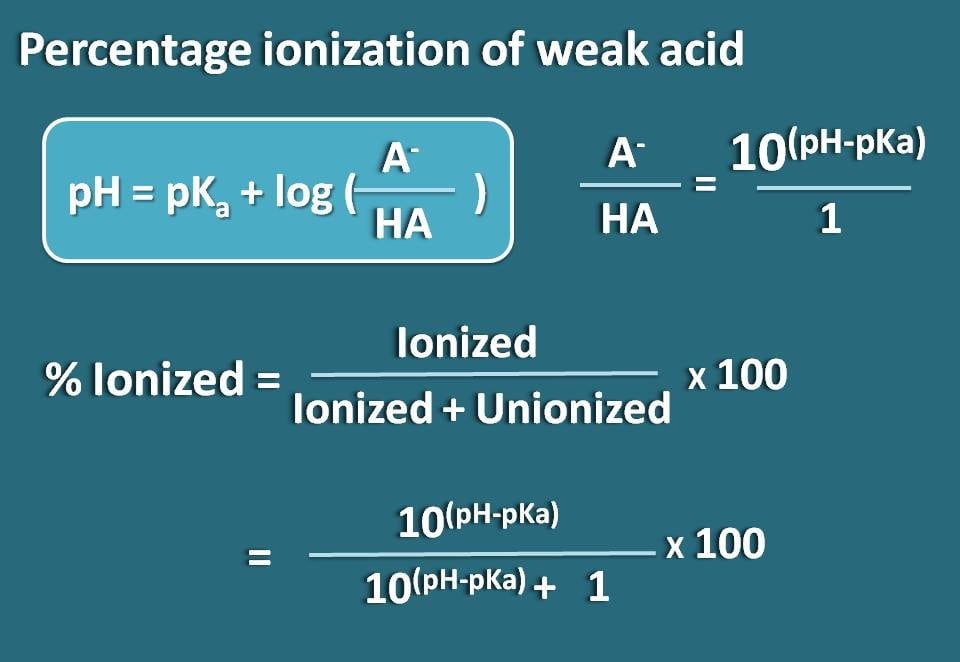
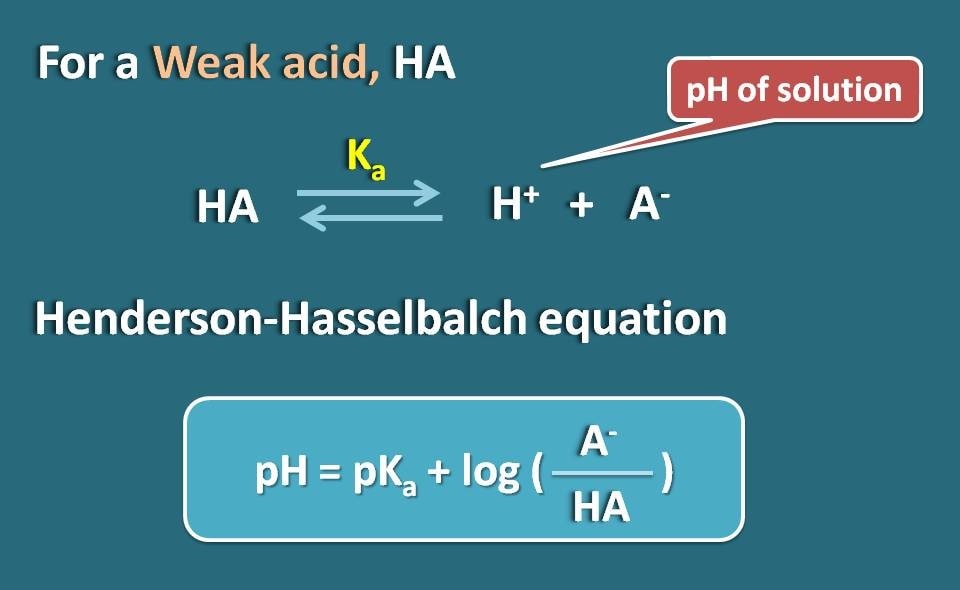
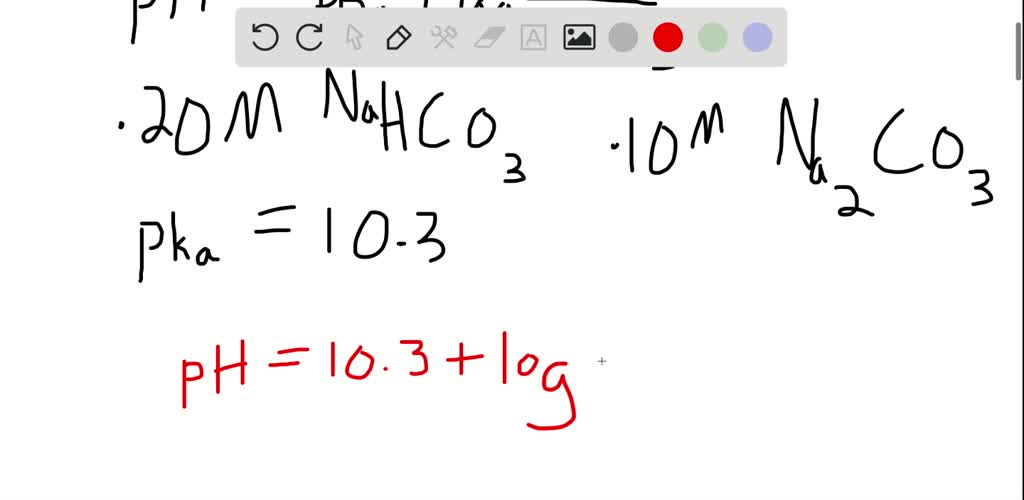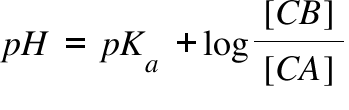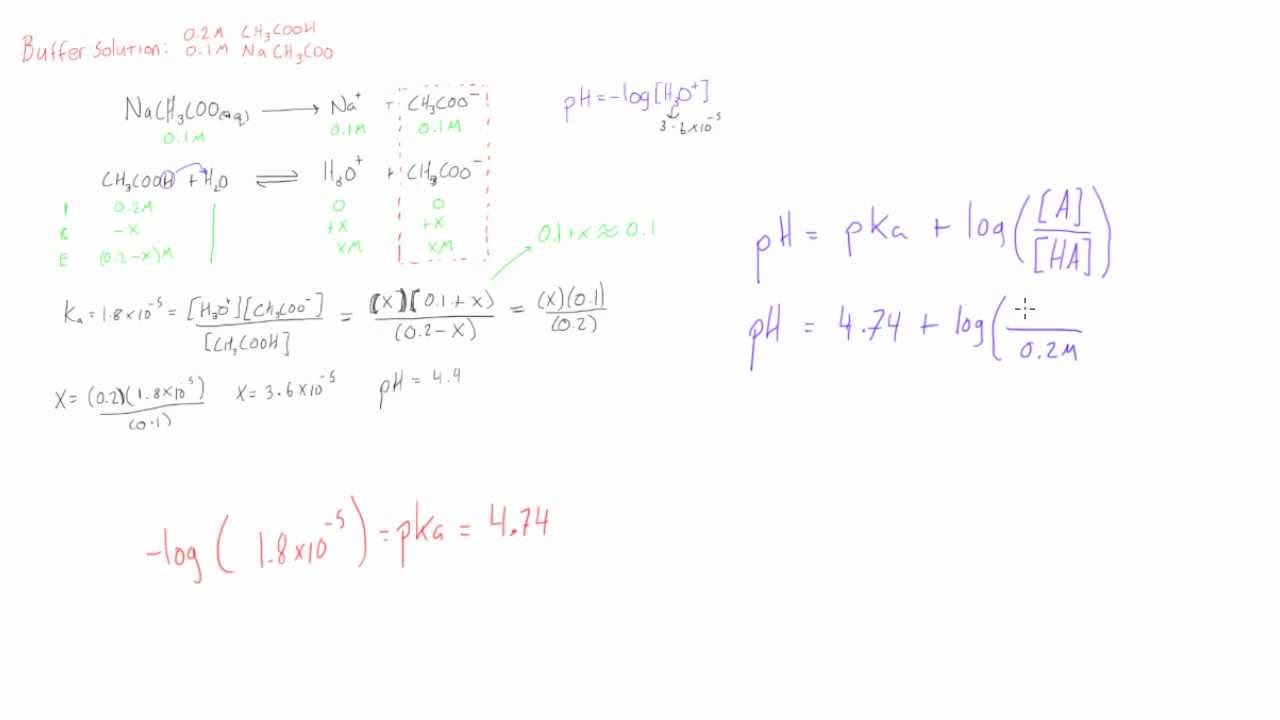Calculating Blood pH-- The Henderson-Hasselbalch Equation - Anatomy & Physiology Video | Clutch Prep
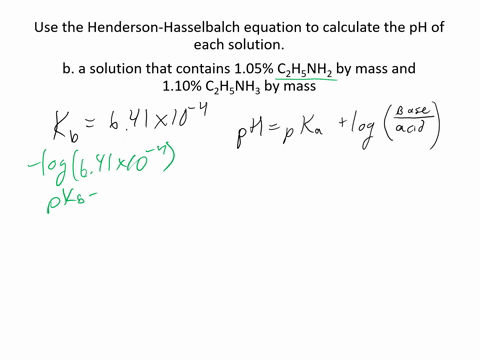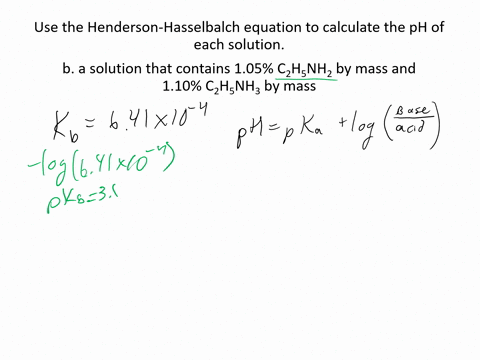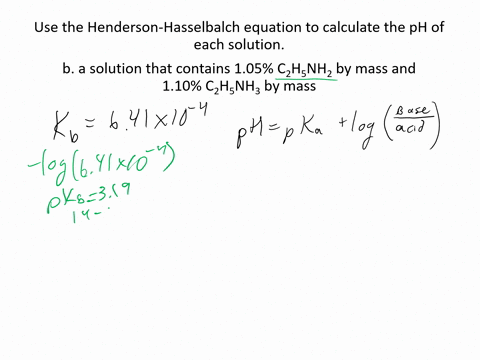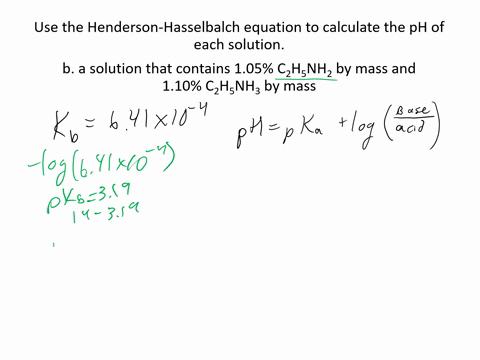
SOLVED:Use the Henderson-Hasselbalch equation to calculate the pH of each solution. a. a solution that is 0.135 M in HClO and 0.155 MinKClO b. a solution that contains 1.05 %C2H5NH2 by mass

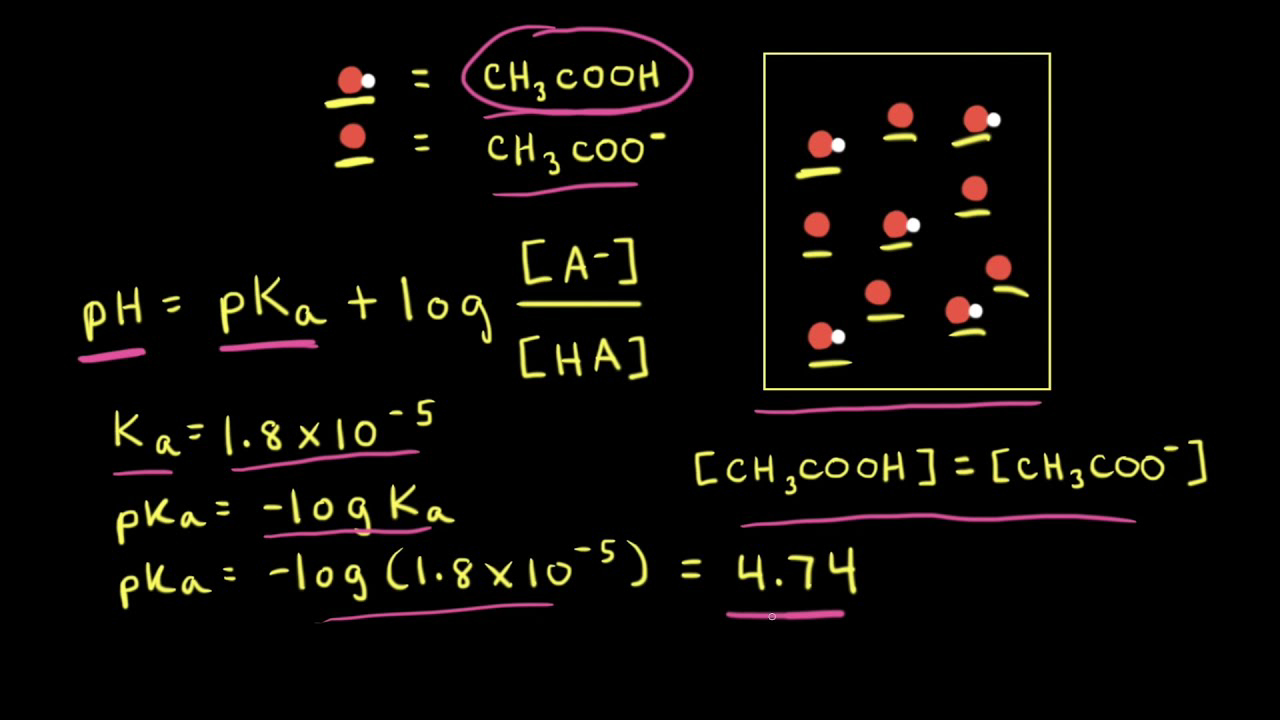
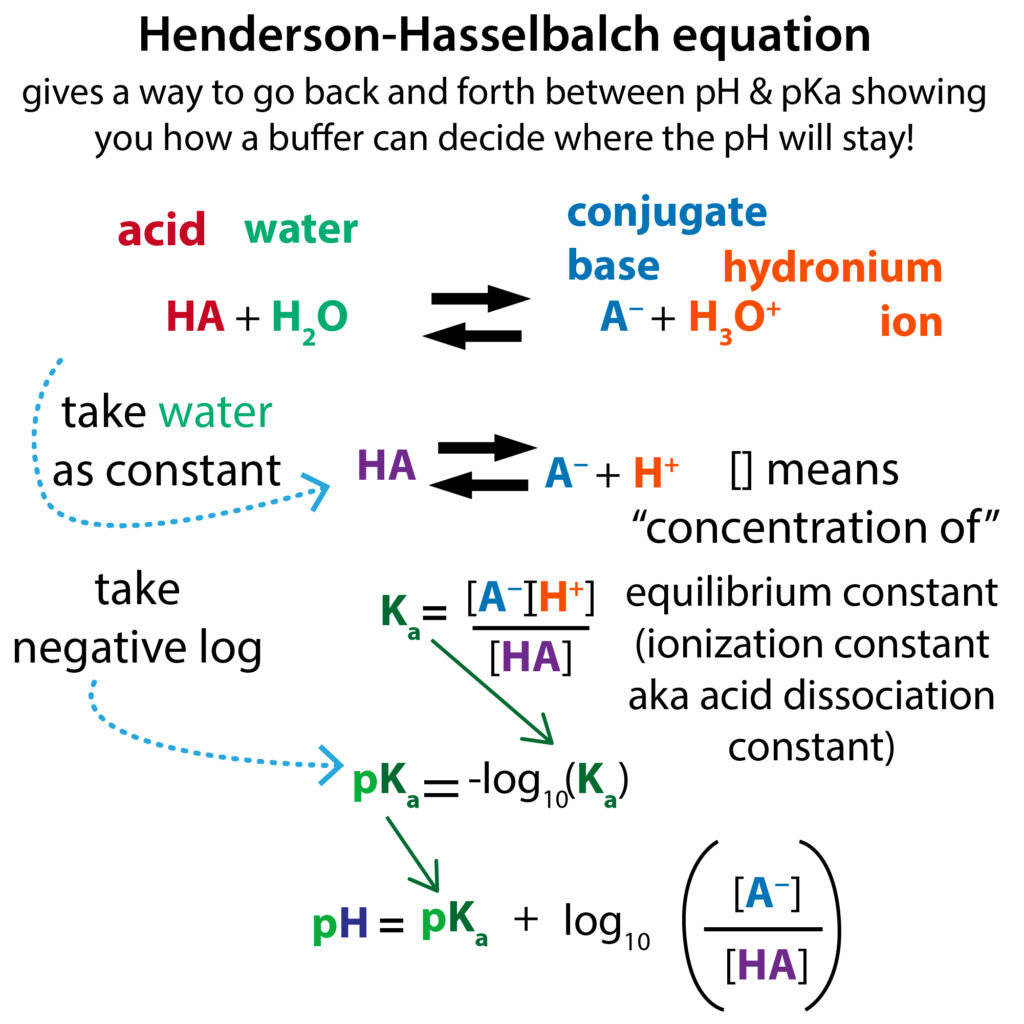
Applications and Example Problems Using Henderson–Hasselbalch Equation | Analytical Chemistry | PharmaXChange.info