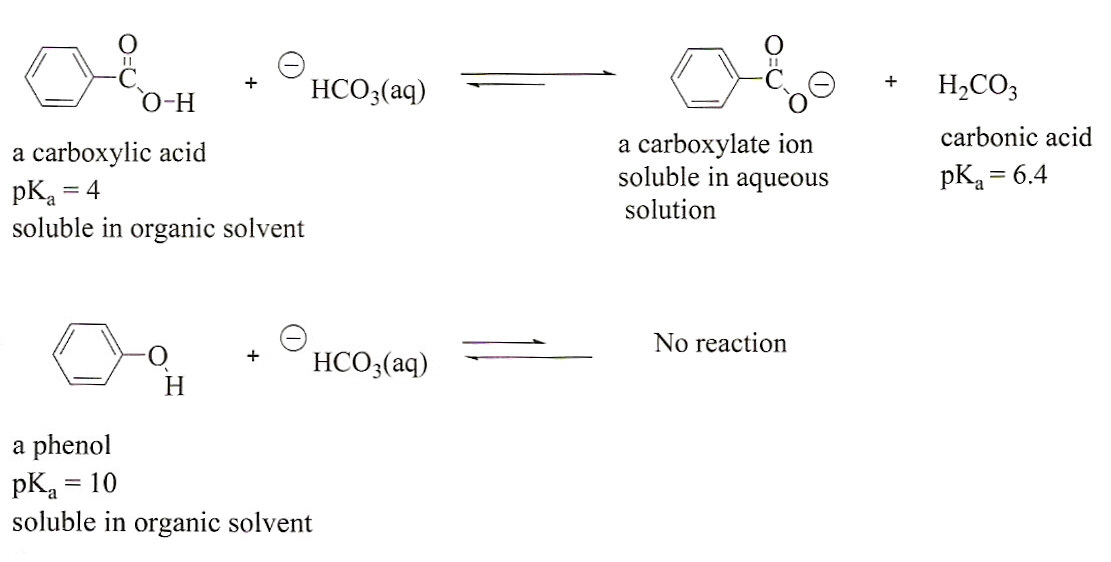
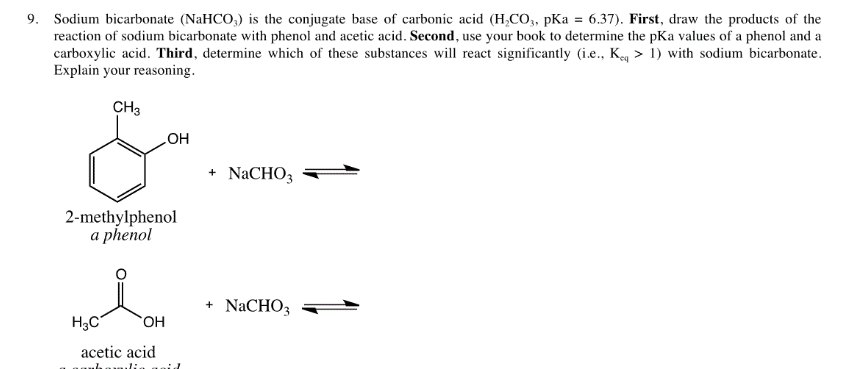
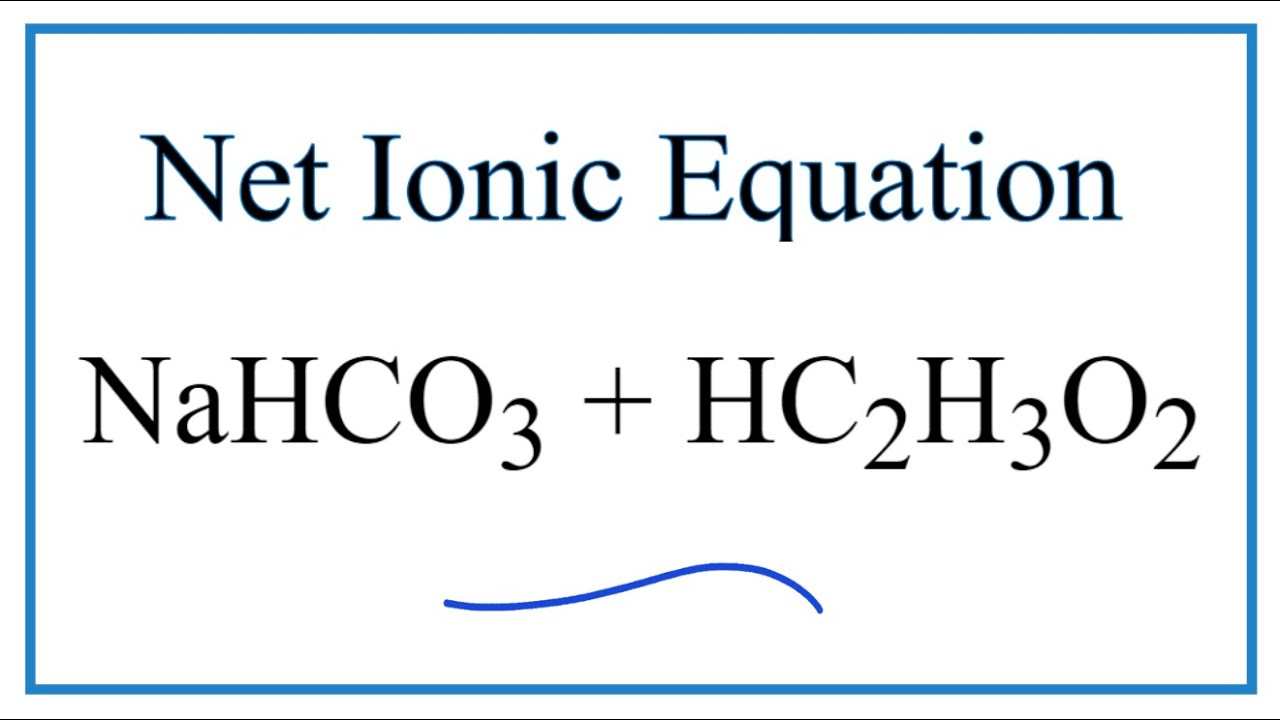Using aqueous hydrochloric acid, sodium bicarbonate, or sodium hydroxide solutions, devise a flow-chart separation scheme to separate the following two-component mixtures. Both substances are soluble | Homework.Study.com

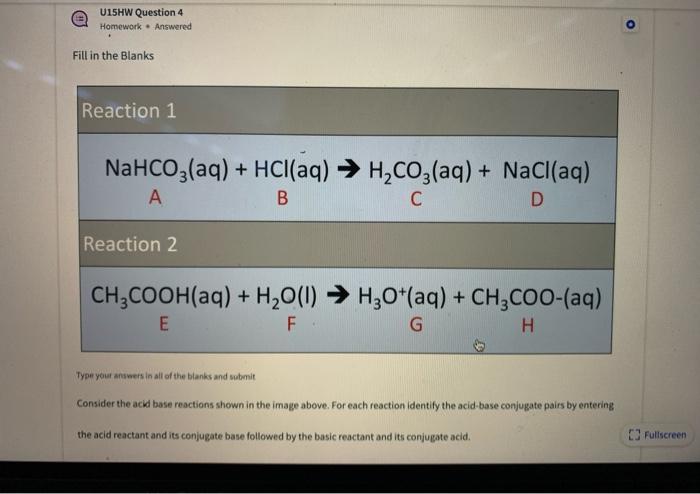
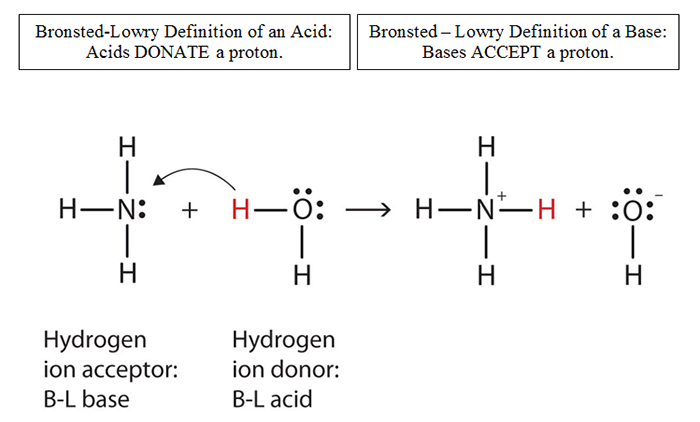
SOLVED: Consider sodium bicarbonate, NaHCOg' also known as baking soda, which is water-soluble, amphiprotic salt with K 4.8x10 and Kb 2.4x10 Which chemical equation is not something that happens when sodium bicarbonate

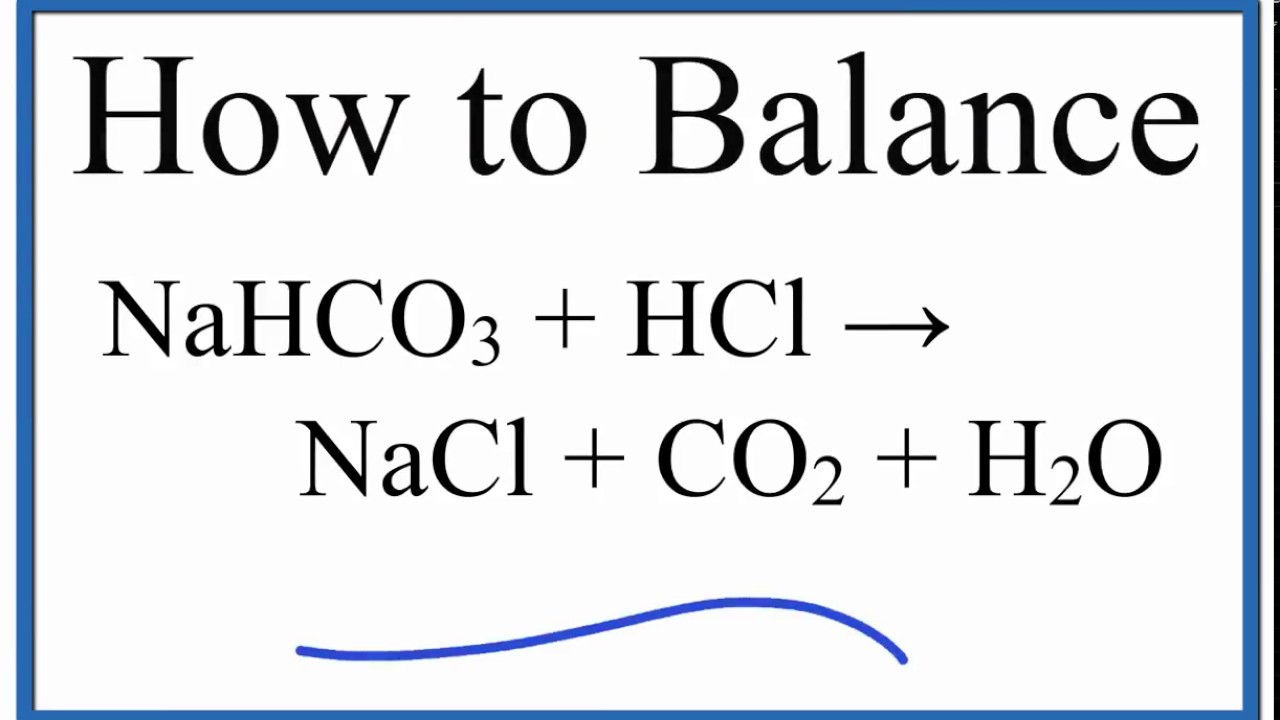
How to Balance NaHCO3 + HCl = NaCl + CO2 + H2O (sodium bicarbonate plus hydrochloric acid) - YouTube
72.Baking soda is an acidic salt or a basic salt.? (NaHCO3 is formed from a weak acid and a strong base so it should be basic but it also has one replaceable
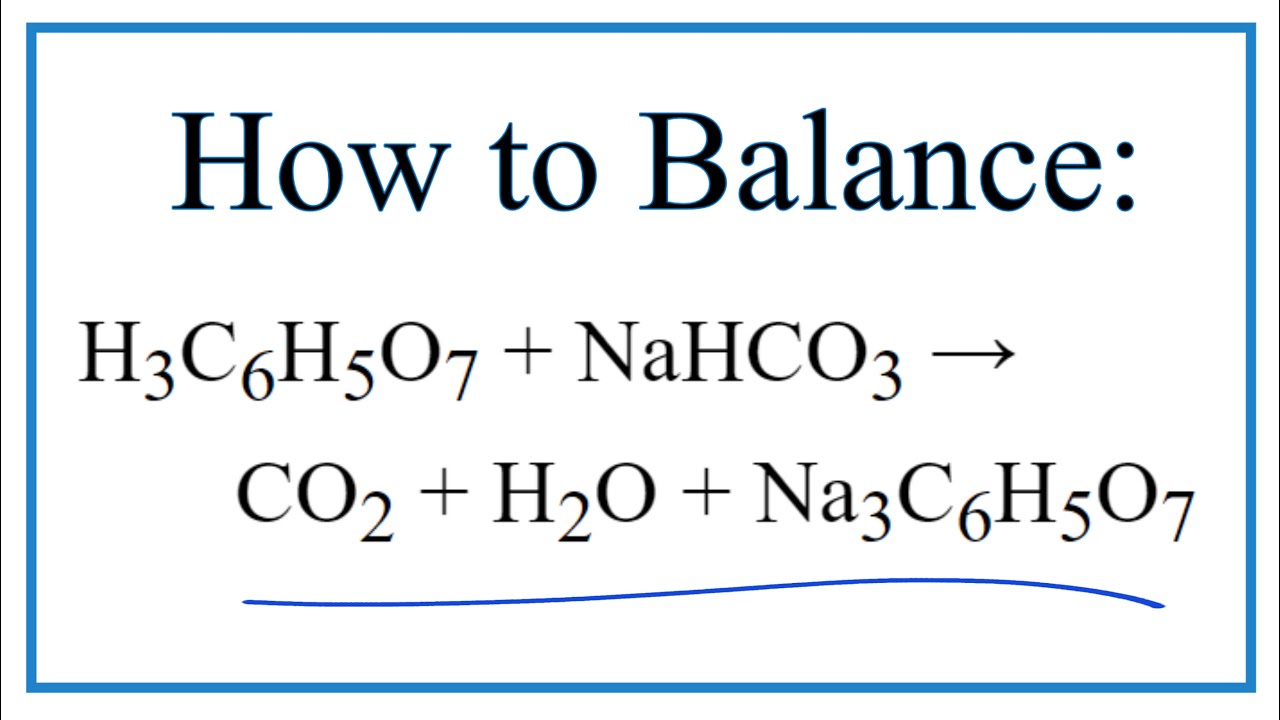
How to Balance H3C6H5O7 + NaHCO3 = CO2 + H2O + Na3C6H5O7 (Citric acid + Sodium bicarbonate ) - YouTube