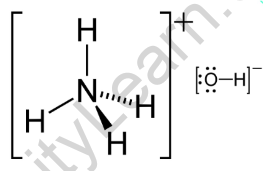
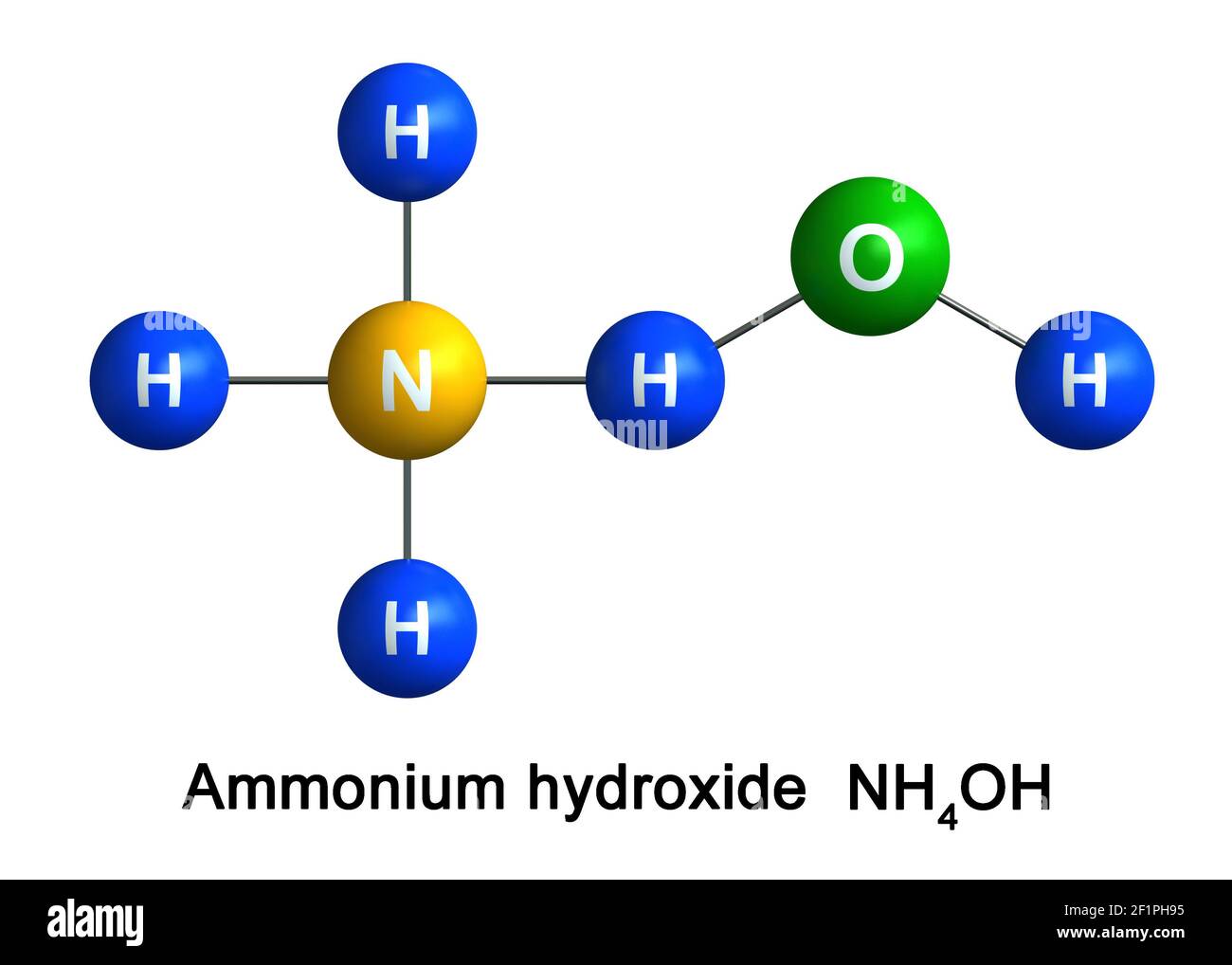
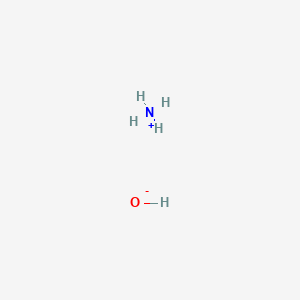
Question Video: The Net Ionic Equation for the Neutralization Reaction between Ammonium Hydroxide and Hydrochloric Acid | Nagwa

Catalytic Alkynylation of Ketones and Aldehydes Using Quaternary Ammonium Hydroxide Base | The Journal of Organic Chemistry

Reaction of 1 with NH4OH in DMSO. (a) UV-vis spectral observations in... | Download Scientific Diagram
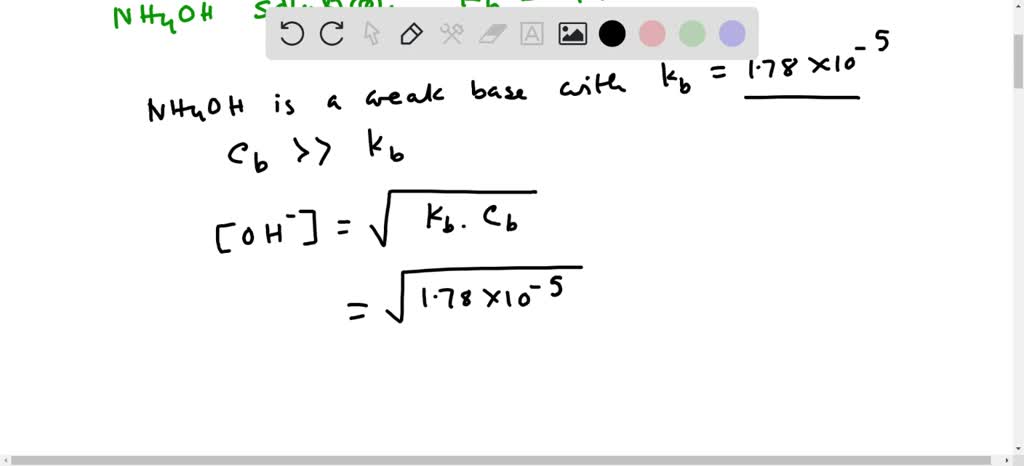


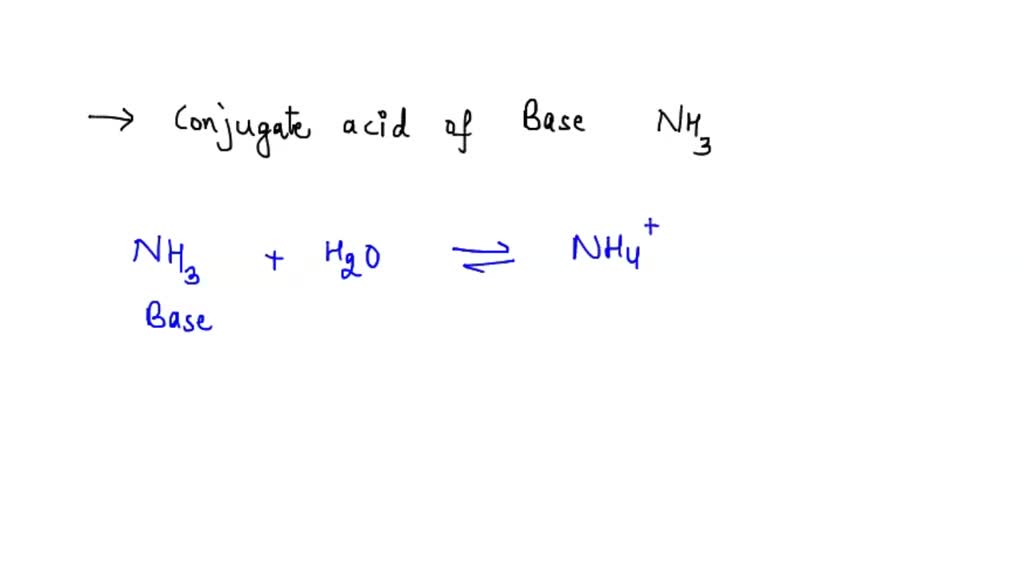



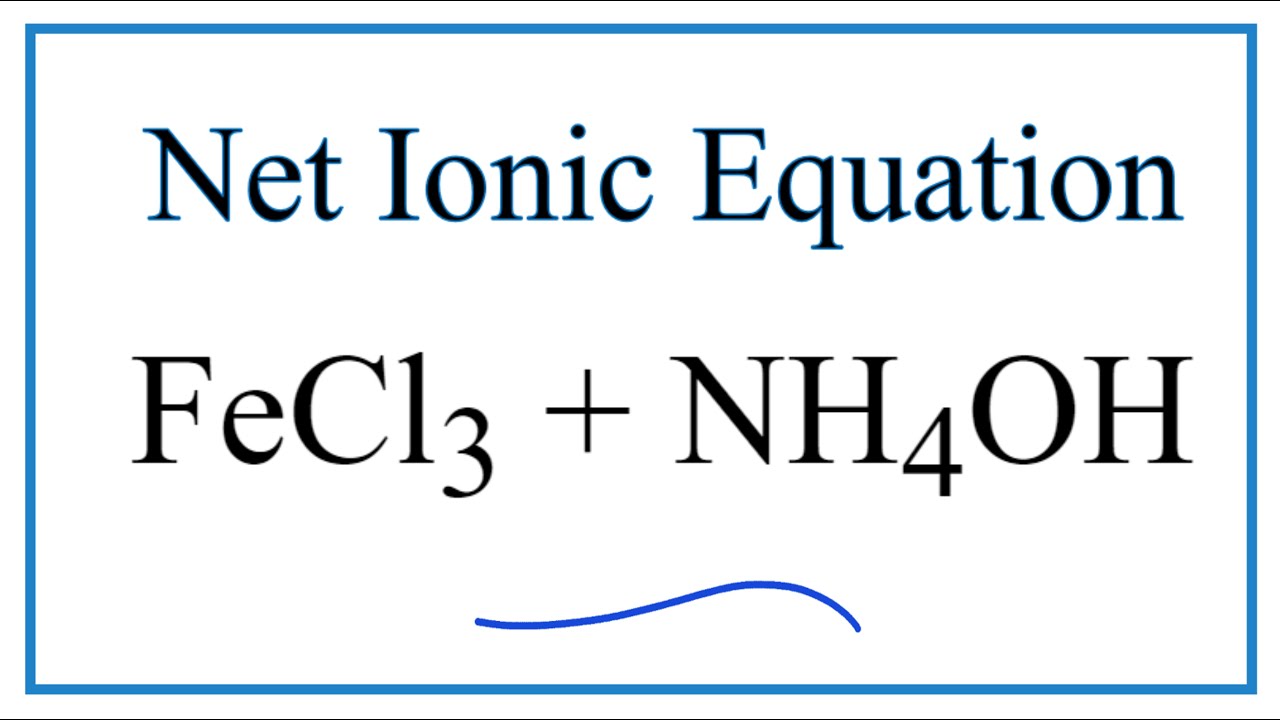


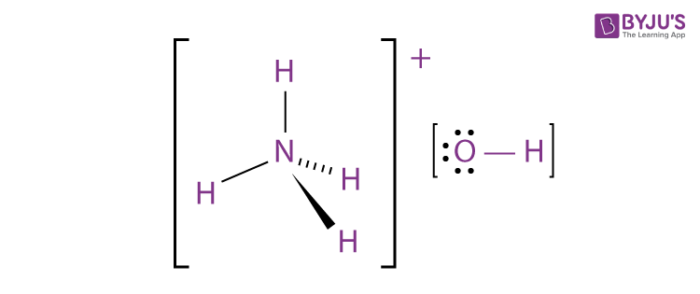


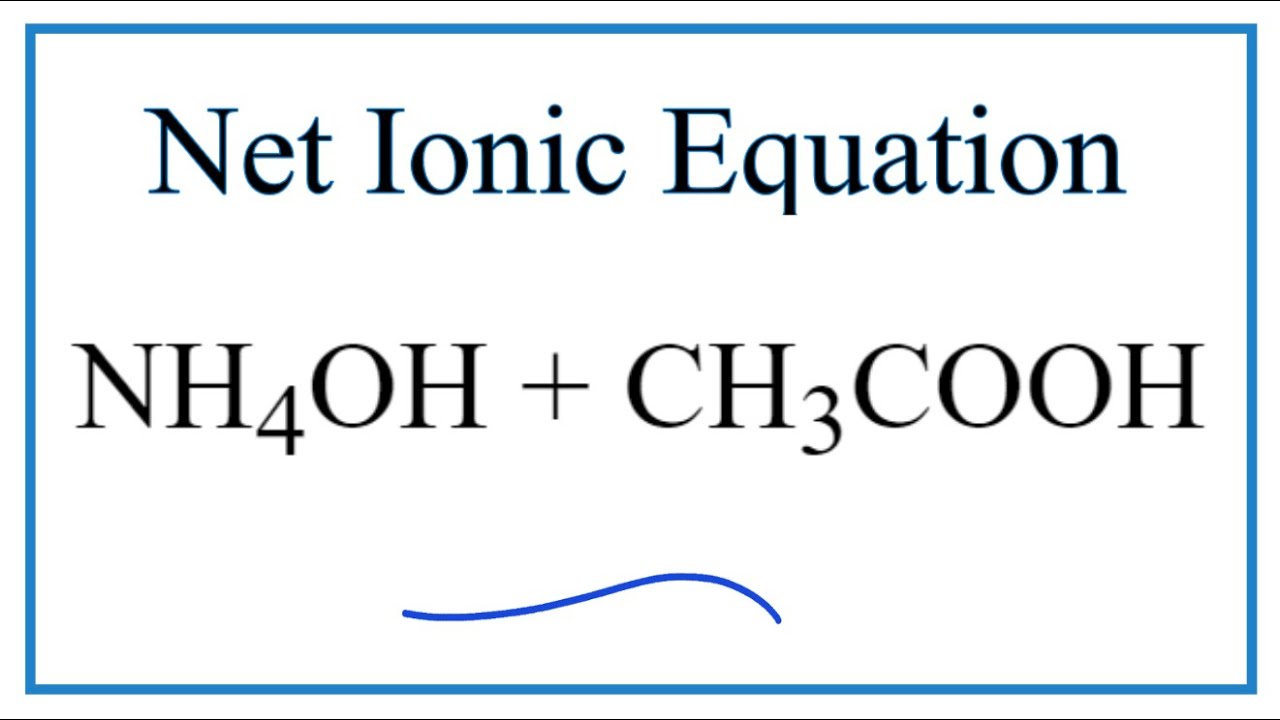

![ANSWERED] When aqueous solutions of NH4OH(aq) and Cu... - Organic Chemistry ANSWERED] When aqueous solutions of NH4OH(aq) and Cu... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/52914656-1658983365.4892888.jpeg)